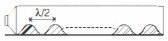
A closed pipe of length $300 \,cm$ contains some sand. A speaker is connected at one of its ends. The frequency of the speaker at which the sand will arrange itself in $20$ equidistant piles is close to .......... $kHz$ (velocity of sound is $300 \,m / s )$

KVPY 2017, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Which of the following is $CORRECT$View Solution
- 2A person is producing wave in string by moving his hand first up and then down. If frequency is $\frac{1}{8}\,Hz$ then find out time taken by particle which is at a distance of $9\,m$ from source to move to lower extreme first time .... $s$ . (Given $\lambda = 24\, m$)View Solution
- 3View SolutionIn a standing wave on a string rigidly fixed at both ends
- 4A wave represented by the given equation $y = a\cos (kx - \omega \,t)$ is superposed with another wave to form a stationary wave such that the point $x = 0$ is a node. The equation for the other wave isView Solution
- 5The power of a sound from the speaker of a radio is $20 mW$. By turning the knob of the volume control, the power of the sound is increased to $400 mW.$ The power increase in decibels as compared to the original power is .... $dB$View Solution
- 6When two sound waves with a phase difference of $\pi /2$, and each having amplitude $A$ and frequency $\omega $, are superimposed on each other, then the maximum amplitude and frequency of resultant wave isView Solution
- 7A student is performing an experiment using a resonance column and a tuning fork of frequency $244 s ^{-1}$. He is told that the air in the tube has been replaced by another gas (assume that the column remains filled with the gas). If the minimum height at which resonance occurs is $(0.350 \pm 0.005) m$, the gas in the tube isView Solution
(Useful information) : $\sqrt{167 R T}=640 j^{1 / 2} mole ^{-1 / 2} ; \sqrt{140 RT }=590 j ^{1 / 2} mole ^{-1 / 2}$. The molar masses $M$ in grams are given in the options. Take the value of $\sqrt{\frac{10}{ M }}$ for each gas as given there.)
- 8The relation between time and displacement for two particles is given byView Solution
${y_1} = 0.06\sin 2\pi (1.04t + {\phi _1})$,
${y_2} = 0.03\sin 2\pi (1.04t + {\phi _2})$
The ratio of the intensity of the waves produced by the vibrations of the two particles will be
- 9A whistle giving out $450 Hz$ approaches a stationary observer at a speed of $33 m/s.$ The frequency heard by the observer in $Hz$ isView Solution
- 10A wire of density $9 \times 10^{-3} \,kg\, cm ^{-3}$ is stretched between two clamps $1\, m$ apart. The resulting strain in the wire is $4.9 \times 10^{-4}$. The lowest frequency of the transverse vibrations in the wire is......$HZ$View Solution
(Young's modulus of wire $Y =9 \times 10^{10}\, Nm ^{-2}$ ), (to the nearest integer),