Assume that a particular amino acid has an isoelectric point of $6.0.$ In a solution at $pH \,1.0$ which of the following species will predominate ?
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe total number of distinct naturally occurring amino acids obtained by complete acidic hydrolysis of the peptide shown below is
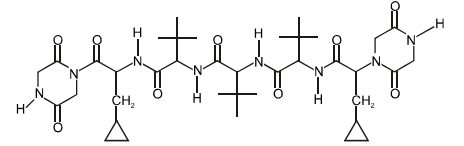
- 2View SolutionWhich one is a disaccharide
- 3View SolutionWhich of the following is not a fat soluble vitamin?
- 4Molecular formula of pentahydroxy acid obtained when glucose is oxidised with $B{r_2}$ water isView Solution
- 5View SolutionThe secondary structure of a protein refers to
- 6View SolutionIdentify the basic amino acid
- 7When the following aldohexose exists in its $D$-configuration, the total number of stereoisomers in its pyranose form is :View Solution
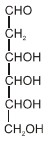
- 8View SolutionThe calorific value is maximum in case of
- 9View SolutionWhich of the following is non-reducing sugar?
- 10$D$ -glucose and $L$ -glucose areView Solution
