Describe the structure of a fuel cell. Draw labelled diagram of a structure of fuel cell?
(Sr. Sec. Board Exam., 2023)
Fuel cells : The galvanic cell in which fuels like hydrogen $\left( H _2\right)$, methane $\left( CH _4\right)$, and methanol $\left( CH _3 OH \right)$ are present whose combustion energy is directly converted in to electrical energy which is called fuel cell. In the most successful fuel cell, the reaction of formation of water by the action of hydrogen and oxygen has been used.
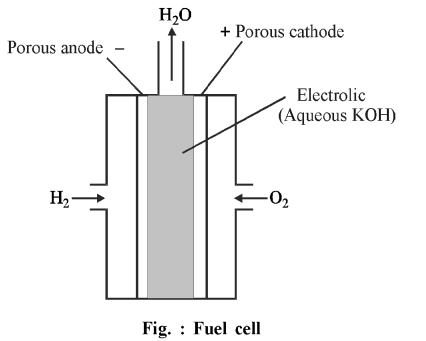
In the fuel cell, bubbles of $H _2$ and $O _2$ gases flow in to the concentrated aqueous NaOH solution through porous carbon electrodes. To increase the speed of electrode reaction, finely divided platinum or palladium metal (catalyst) is mixed in to the electrodes. Electrode reactions are as follows:
Cathode: $O _2(g)+2 H _2 O (l)+4 e ^{-} \rightarrow 4 OH ^{-}( aq )$
Anode: $2 H _2(g)+4 OH ^{-}( aq ) \rightarrow 4 H _2 O ( l )+4 e ^{-}$
The net reaction can be written as :
$
2 H_2(g)+O_2(g) \rightarrow 2 H_2 O(l)
$
As long as $H _2$ and $O _2$ gas continues to flow, electricity continues to be obtained, hence this cell never ends. Thermal plants have an efficiency of only $40 \%$ while fuel cells have an efficiency of $70 \%$.
Fuel cells were used to provide electricity in Apollo space program and water vapours formed by the reaction of $H _2$ and $O _2$ was condensed and used as drinking water for astronauts.
New electrode materials, catalysts and good electrolytes have been developed to increase the efficiency of fuel cells.
Due to being pollution free, fuel cells have been used in vehicles on trial basis.

In the fuel cell, bubbles of $H _2$ and $O _2$ gases flow in to the concentrated aqueous NaOH solution through porous carbon electrodes. To increase the speed of electrode reaction, finely divided platinum or palladium metal (catalyst) is mixed in to the electrodes. Electrode reactions are as follows:
Cathode: $O _2(g)+2 H _2 O (l)+4 e ^{-} \rightarrow 4 OH ^{-}( aq )$
Anode: $2 H _2(g)+4 OH ^{-}( aq ) \rightarrow 4 H _2 O ( l )+4 e ^{-}$
The net reaction can be written as :
$
2 H_2(g)+O_2(g) \rightarrow 2 H_2 O(l)
$
As long as $H _2$ and $O _2$ gas continues to flow, electricity continues to be obtained, hence this cell never ends. Thermal plants have an efficiency of only $40 \%$ while fuel cells have an efficiency of $70 \%$.
Fuel cells were used to provide electricity in Apollo space program and water vapours formed by the reaction of $H _2$ and $O _2$ was condensed and used as drinking water for astronauts.
New electrode materials, catalysts and good electrolytes have been developed to increase the efficiency of fuel cells.
Due to being pollution free, fuel cells have been used in vehicles on trial basis.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionExplain the mechanism of rusting of iron.
- 2View Solution(a) Draw the structure of a galvanic cell and also describe the reaction taking place in it.
(b) What happens when opposite external potential is applied in the galvanic cell system? Explain. - 3Derive the Nernst equation and explain how $E_{\text {cell }}^{\circ}$ depends on concentration of ions.View Solution
- 4View SolutionExplain fuel cell in detail and explain the shortcomings of power generation from thermal plants.
- 5Explain the relationship between $\Delta G$ and $E _{\text {cell }}^{\circ}$ with the help of an example.View Solution
- 6View SolutionDescribe the standard hydrogen electrode with diagram.
- 7Explain relationship between $E _{\text {cell }}^{\circ}$ and equilibrium constant with the help of Nernst equation.View Solution
- 8View SolutionWhat is called a battery? Explain its characteristics and describe dry cell and mercury cell.
- 9View SolutionDescribe graphically the experimental method of measuring the conductivity of electrolyte solutions (ionic solution).
