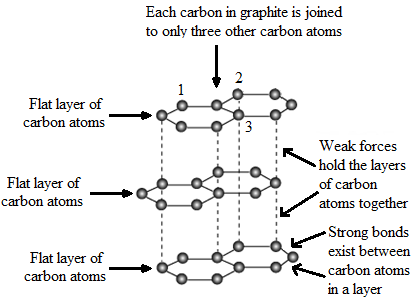
Describe the structure of graphite with the help of a labelled diagram.
The structure of graphite is very different from that of diamond. A graphite crystal consists of layers of carbon atoms or sheets of carbon atoms. Each carbon atom in a graphite layer is joined to other three carbon atoms by strong covalent bonds to form flat hexagonal rings. The various layers of carbon atoms in graphite are held together by weak Van der Waals forces. Due to this sheet like structure, graphite is a comparatively soft substance.

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1You are given the following molecular formulae of some hydrocarbons:View Solution
$C _5 H _8 ; C _7 H _{14} ; C _6 H _6 ; C _5 H _{10} ; C _7 H _{12} ; C _6 H _{12}$
a. Which formula represent cyclohexane as well as hexene?
b. Which formula represents benzene?
c. Which three formulae represent open chain unsaturated hydrocarbons having double bonds?
d. Which two formulae represent unsaturated hydrocarbons having triple bonds?
e. Which three formulae can represent cyclic hydrocarbons? - 2An organic compound $A$ having the molecular formula $C _3 H _8 O$ is a liquid at room temperature. The organic liquid $A$ reacts with sodium metal to evolve a gas which burns causing a little explosion. When the organic liquid A heated with concentrated sulphuric acid at $170^{\circ} C$, it forms a compound B which decolourizes bromine water. The compound B adds on one molecule of hydrogen in the presence of Ni as catalyst to forms compound C which gives substitution reactions with chlorine.View Solution
a. What is compound A ?
b. What is compound B ?
c. What type of reaction occurs when A is converted into B ?
d. What is compound C?
e. What type of reaction takes place when $B$ is converted into $C$ ? - 3View SolutionDescribe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
- 4You are given an organic compound having the molecular formula $C _3 H _8$. Give the name and formula of the compound formed:View Solution
a. When one H atom of $C _3 H _8$ is replaced by a Cl atom.
b. When one H atom of $C _3 H _8$ is replaced by a OH group.
c. When one H atom of $C _3 H _8$ is replaced by a CHO group.
d. When one H atom of $C _3 H _8$ is replaced by a COOH group.
e. When two H atoms joined to the middle carbon atom of $C _3 H _8$ are replaced by one O atom. - 5Consider the following organic compounds:View Solution
$CH _3 OH , C _2 H _5 OH , CH _3 COCH _3, CH _3 COOH , C _2 H _5 COOH , C _4 H _9 COOC _2 H _5, CH _4, C _2 H _6, CH _3 CHO , HCHO$
Out of these compounds:
a. Which compound is most likely to be sweet-smelling?
b. Which compound on treatment with conc. $H _2 SO _4$ at $170^{\circ} C$ forms an alkene?
c. Which compound on repeated chlorination forms chloroform?
d. Which compound is added to alcohol to denature it?
e. Which compound is a constituent of vinegar?
f. Which compound is used to sterilise wounds and syringes? - 6A colourless organic liquid $X$ of molecular formula $C _2 H _4 O _2$ turns blue litmus to red. Another colourless organic liquid Y of molecular formula $C _3 H _6 O$ has no action on any litmus but it is used as a nail polish remover. A yet another colourless organic liquid Z of molecular formula $C _2 H _6 O$ has also no action on litmus but it is used in tincture of iodine.View Solution
a. Name the liquid X . To which homologous series does it belong? Give the name of another member of this homologous series.
b. Name the liquid Y. To which homologous series does it belong? Write the name of another member of this homologous series.
c. Can you name an organic compound having the same molecular formula as liquid $Y$ but which belongs to a different homologous series? What is this homologous series?
d. Name the liquid Z. To which homologous series does it belong? Write the name of another member of this homologous series. - 7View SolutionThe solid element A exhibits the property of catenation. It is also present in the form of a gas B in the air which is utilised by plants in photosynthesis. An allotrope C of this element is used in glass cutters.
- What is element A?
- What is the gas B?
- Name the allotrope C.
- State another use of allotrope C (other than in glass cutters).
- Name another allotrope of element A which exists as spherical molecules.
- Name a yet another allotrope of element A which conduct electricity.
- 8A four carbons atoms containing neutral organic compound $X$ reacts with sodium metal to evolve a gas which burns with 'pop' sound. Another four carbon atoms containing carbon compound reacts with sodium hydrogen carbonate to evolve a gas which turns lime water milky. When compounds $X$ and $Y$ are heated together in the presence of a little of concentrated sulphuric acid, then a new compound $Z$ is formed.View Solution
a. What is compound $X$ ? Also write its formula.
b. What is compound Y? Also write its formula.
c. What is compound Z? Also write its formula.
d. What type of smell is given by compound Z?
e. What is the general name of compounds like $Z$ ?
f. What is the general name of the reaction which which takes place between $X$ and $Y$ to form $Z$ ? - 9View SolutionExplain the cleansing action of soap. Draw diagrams to illustrate your answer.
- 10An organic compound A (molecular formula $C _2 H _4 O _2$ ) reacts with Na metal to form a compound B and evolves a gas which burns with a pop sound. Compound A on treatment with an alcohol C in the presence of a little of concentrated sulphuric acid forms a sweet-smelling compound D (molecular formula $C _3 H _6 O _2$ ). Compound D on treatment with NaOH solution gives back B and C. Identify A, B, C and D.View Solution
