For a double strand $DNA$, one strand is given below:
The amount of energy required to split the double strand $DNA$ into two single strands is . . . .kcal $mol ^{-1}$.
[Given: Average energy per $H$-bond for $A - T$ base pair $=1.0 kcal mol ^{-1}, G - C$ base pair $=1.5 kcal mol ^{-1}$, and $A - U$ base pair $=1.25 kcal mol ^{-1}$. Ignore electrostatic repulsion between the phosphate groups.]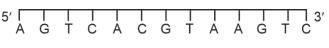
IIT 2024, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following vitamin is water soluble?
- 2Identify the correct set of stereochemical relationships amongst the following monosaccharides $I-IV$View Solution

- 3$pH$ at isoeletric point of three amino acids are given as follow.View Solution
$pH$ at Isoelectric point $(Amino acid)_I$ $6$ $(Amino acid)_{II}$ $9.8$ $(Amino acid)_{III}$ $3.9$ $(Amino acid)_{II}$ is ____ ?
- 4View SolutionProteins are built up of
- 5View SolutionA certain compound gives negative test with ninhydrin and positive test with Benedict's solution. The compound is
- 6A sugar ' $X$ ' dehydrates very slowly under acidic condition to give furfural which on further reaction with resorcinol gives the coloured product after sometime. Sugar ' $X$ ' isView Solution
- 7View SolutionExample of aromatic amino acid is/are
- 8View SolutionThe enzyme ptylin used for the digestion of food is present in
- 9View SolutionThe example of a protein is
- 10View SolutionWhich of the following gives a positive test with ninhydrin?
