Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The correct decreasing order of dehydration of following alcohols with conc. $\ce{H_{2}SO_{4}}$ is$:$View Solution
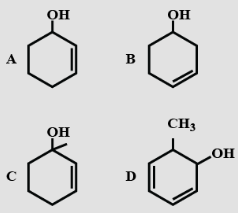
- 2View SolutionWhich of the following phenols has lowest solubility in water?
- 3The compound which gets dissolved in water is$:$View Solution
- 4View Solution
- 5View SolutionEthers show dipolar nature due to the presence of ______.
- 6$\ce{IUPAC}$ name of $m-$cresol is $.......$View Solution
- 7View SolutionPrimary, secondary and tertiary alcohols can be distinguish by:
- 8View SolutionWhich of the following reactions is possible?
- 9View SolutionWhich of the following reacts fastly with Na?
- 10View SolutionWhich of the following can not be the product of this reaction?
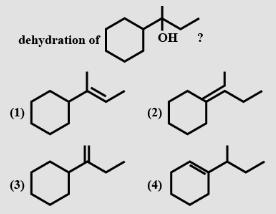
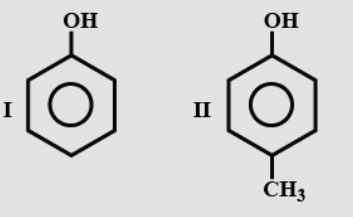

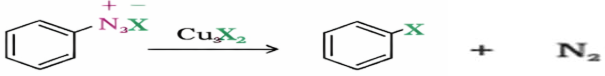 is-
is-