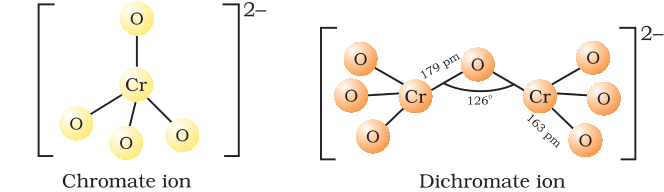
The ionic charges on chromate ion and dichromate ion respectively is:
- -2, -2
Chromate salts contain the chromate anion, $\text{CrO}^{2-}_4$ with -2 ionic charge.
Dichromate salts contain the dichromate anion, $\text{CrO}^{2-}_7$ with -2 ionic charge.
They are oxoanions of chromium in the +6 oxidation state.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following becomes paramagnetic on heating?
- 2View SolutionColour in transition metal compounds is attributed to:
- 3View SolutionVariable valency is characteristic of:
- 4View SolutionTransition metals show paramagnetism due to:
- 5View SolutionIt is sometimes necessary to remove colouring matter contained as an impurity in glass. Glass is decolourised by:
- 6View SolutionWhich forms coloured salts?
- 7Number of Cr−O bonds in dichromate ion $(\text{Cr}_2\text{O}_7^{2-})$ is:View Solution
- 8View SolutionWhich of the following has highest value of magnetic moment?
- 9View SolutionOut of the following transition elements, the maximum number of oxidation states are shown by:
- 10View SolutionThe first ionisation potential of N, P, O and S are in the order of:
