The specific rotation of equilibrium mixture of $\alpha - D-$ glucose and $\beta - D-$ glucose, is ....... $^o$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo monomers in maltose are
- 2View SolutionGlucose has functional group
- 3View SolutionThe pair of compounds in which both the compounds give positive test with Tollen's reagent is
- 4View SolutionEnzymes are
- 5Which one of the following bases is not present in $DNA$ ?View Solution
- 6View SolutionHydrolysis of sucrose with dilute aqueous sulphuric acid yields
- 7View SolutionSucrose is
- 8Match List $I$ with List $II$View Solution
List $I$ List $II$ $A$ $\alpha$-Glucose and $\alpha-$Galactose $I$ Functional isomers $B$ $\alpha$-Glucose and $\beta-$-Galactose $II$ Homologous $C$ $\alpha$-Glucose and $\alpha$-Galactose $III$ Anomers $D$ $\alpha$-Glucose and $\alpha$-Galactose $IV$ Epimers Choose the correct answer from the options given below:
- 9View SolutionThe correct sequence of amino acids present in the tripeptide given below is
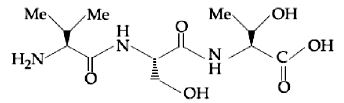
- 10View SolutionInsulin has ......... amino acid
