To determine the Young's modulus of a wire, the formula is $Y = \frac{FL}{A\Delta L};$ where $L$ = length, $A = $area of cross-section of the wire, $\Delta L = $change in length of the wire when stretched with a force $F$. The conversion factor to change it from $CGS$ to $MKS$ system is .............. $10^{-1}\mathrm{N/m}^{2}$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
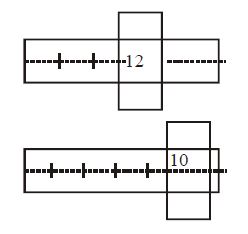
- 1A screw gauge has some zero error but its value is unknown. We have two identical rods. When the first rod is inserted in the screw, the state of the instrument is shown by diagram $(I).$ When both the rods are inserted together in series then the state is shown by the diagram $(II).$ What is the zero error of the instrument ? .......... $mm$View Solution
$1\,M.S.D. = 100\, C.S.D. = 1\, mm $
- 2$SI$ unit of pressure isView Solution
- 3A massive black hole of mass $m$ and radius $R$ is spinning with angular velocity $\omega$. The power $P$ radiated by it as gravitational waves is given by $P=G c^{-5} m^{x} R^{y} \omega^{z}$, where $c$ and $G$ are speed of light in free space and the universal gravitational constant, respectively. Then,View Solution
- 4The length, breath and thickness of a metal sheet are $4.234 \;m , 1.005\; m$, and $2.01\; cm$ respectively then the volume of the sheet isView Solution
- 5The entropy of any system is given byView Solution
${S}=\alpha^{2} \beta \ln \left[\frac{\mu {kR}}{J \beta^{2}}+3\right]$
Where $\alpha$ and $\beta$ are the constants. $\mu, J, K$ and $R$ are no. of moles, mechanical equivalent of heat, Boltzmann constant and gas constant repectively. [Take ${S}=\frac{{dQ}}{{T}}$ ]
Choose the incorrect option from the following:
- 6View SolutionUnit of impulse is
- 7Turpentine oil is flowing through a tube of length $l$ and radius $r$. The pressure difference between the two ends of the tube is $P .$ The viscosity of oil is given by $\eta=\frac{P\left(r^{2}-x^{2}\right)}{4 v l}$ where $v$ is the velocity of oil at a distance $x$ from the axis of the tube. The dimensions of $\eta$ areView Solution
- 8View SolutionWhich of the following system of units is not based on units of mass, length and time alone
- 9According to Joule's law of heating, heat produced $H = {I^2}\,Rt$, where I is current, $R$ is resistance and $t$ is time. If the errors in the measurement of $I, R$ and t are $3\%, 4\% $ and $6\% $ respectively then error in the measurement of $H$ isView Solution
- 10View SolutionWhich of the following is not a unit of time
