Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
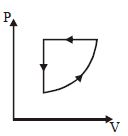
- 2For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
- 3View SolutionEntropy of a thermodynamic system does not change when this system is used for
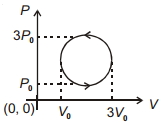
- 4View SolutionWork done in the cyclic process shown in figure is ...........
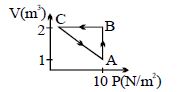
- 5An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
- 6An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 7A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 8View SolutionThe specific heat of a gas at constant pressure is more than that of the same gas at constant volume because
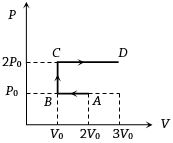
- 9$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
- 10View SolutionIrreversible process is
