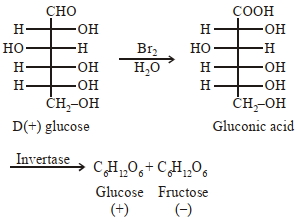
Which of the following statement($s$) is(are) true ?
$(1)$ Oxidation of glucose with bromine water gives glutamic acid
$(2)$ The two six-membered cyclic hemiacetal forms of $D-(+)$-glucose ard called anomers
$(3)$ Hydrolysis of sucrose gives dextrorotatory glucose and laevorotatory fructose
$(4)$ Monosaccharides cannot be hydrolysed to give polyhydroxy aldehydes and ketones
IIT 2019, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA peptide synthesized by the reactions of one molecule each of Glycine, Leucine, Aspartic acid and Histidine will have ..... peptide linkages.
- 2Which of the following is correct structure of $L$ -glucose ?View Solution
- 3View SolutionThe nucleic acid base having two possible binding sites is
- 4The correct match between Item $I$ and Item $II$ isView Solution
Item $I$ Item $II$ $A$ Allosteric effect $P$ Molecule binding to the active site of enzyme $B$ Competitive inhibitor $Q$ Molecule crucial for communication in the body $C$ Receptor $R$ Molecule binding to a site other than the active site of enzyme $D$ Poison $S$ Molecule binding to the enzyme covalently - 5View SolutionWhich of the following is not essential amino acid
- 6The double helical structure of $DNA$ was proposed byView Solution
- 7View SolutionGlucose and Fructose can be differentiated by
- 8View SolutionWhich of the following reagents cannot distinguish between glucose and fructose ?
- 9When sugar ' $X$ ' is boiled with dilute $H _{2} SO _{4}$ in alcoholic solution, two isomers ' $A$ ' and ' $B$ ' are formed. $'A'$ on oxidation with $HNO _{3}$ yields saccharic acid where as ' $B$ ' is laevorotatory. The compound ' $X$ ' is ....... .View Solution
- 10View SolutionOut of the following the best category of proteins is