$0.056 \,kg$ of Nitrogen is enclosed in a vessel at a temperature of $127\,^{\circ} C$. The amount of heat required to double the speed of its molecules is k cal. (Take $R =2$ $cal \,mole$ $^{-1} K ^{-1}$ )
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The average degree of freedom per molecule of a gas is $6$. The gas performs $25\ J$ work, while expanding at constant pressure. The heat absorbed by the gas is .... $J$View Solution
- 2A gas mixture consists of $3\,moles$ of oxygen and $5\,moles$ or argon at temperature $T.$ Considering only translational and rotational modes, the total internal energy of the system isView Solution
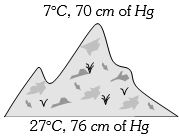
- 3At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 4What is the ratio of specific heats of constant pressure and constant volume for $N{H_3}$View Solution
- 5$E_0$ and $E_H$ respectively represents the average kinetic energy of a molecule of oxygen and hydrogen. If the two gases are at the same temperature, which of the following statement is true?View Solution
- 6The temperature of $5$ moles of a gas which was held at constant volume was changed from ${100^o}C$ to ${120^o}C$. The change in internal energy was found to be $80$ Joules. The total heat capacity of the gas at constant volume will be equal to ...... $J/K$View Solution
- 7View SolutionThe specific heat of an ideal gas is
- 8For a gas $\gamma = 7/5.$ The gas may probably beView Solution
- 9In an ideal gas at temperature $T$, the average force that a molecule applies on the walls of a closed container depends on $T$ as ${T^q }$ . A good estimate for $q$ isView Solution
- 10Two moles of a monoatomic ideal gas is confined in a container and is heated such that its temperature increases by $10\,^oC$. The approximate change in its internal energy is ..... $J$. $(R = 8.31\, J/mole-K)$View Solution
