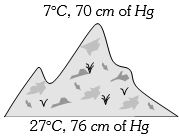
At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom is

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 2In a thermally isolated system, two boxes filled with an ideal gas are connected by a valve. When the valve is in closed position, states of the box $1$ and $2$ respectively, are ( $1 \,atm , V, T)$ and $(0.5 \,atm , 4 V, T)$. When the valve is opened, then the final pressure of the system is approximately ............... $atm$View Solution
- 3If two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats $\gamma=$ ......View Solution
- 4If the degree of freedom of a gas are $f,$ then the ratio of two specific heats ${C_P}/{C_V}$ is given byView Solution
- 5On giving equal amount of heat at constant volume to $1\, mol$ of a monoatomic and a diatomic gas the rise in temperature $(\Delta T)$ is more forView Solution
- 6Two closed containers of equal volume filled with air at pressure $P_0$ and temperature $T_0$. Both are connected by a narrow tube. If one of the container is maintained at temperature $T_0$ and other at temperature $T$, then new pressure in the containers will beView Solution
- 7The correct relation between $\gamma=\frac{ C _p}{ c _v}$ and temperature $T$ is :View Solution
- 8View SolutionAt a given temperature the root mean square velocities of oxygen and hydrogen molecules are in the ratio
- 9If the ratio of vapour density for hydrogen and oxygen is $\frac{1}{{16}}$, then under constant pressure the ratio of their rms velocities will beView Solution
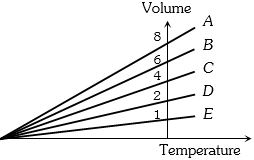
- 10The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2\, m$ at a pressure $2\,P$ is given by the straight lineView Solution