$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
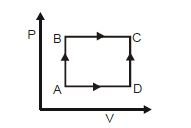
- 1A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
- 2A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
- 3View SolutionThe internal energy of the gas increases In
- 4For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
- 5$n-$ $moles$ of an ideal gas with constant volume heat capacity $C_v$ undergo an isobaric expansion by certain volume. The ratio of the work done in the process, to the heat supplied isView Solution
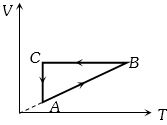
- 6A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 7An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
- 8Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from $2 \times {10^{ - 3}}\,{m^3}$ to $3.34{m^3}.$ The work done by the system is about ....... $kJ$View Solution
- 9Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
- 10An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
