A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
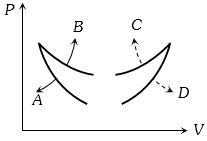
- 1Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
- 2View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 3A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 4View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
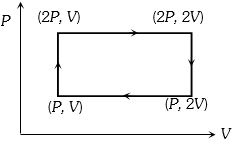
- 5Work done in the given $P-V$ diagram in the cyclic process isView Solution
- 6For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 7A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 8A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
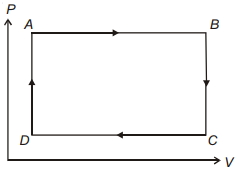
- 9The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
- 10The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
