$1\ mole$ of a gas with $\gamma$ = $7/5$ is mixed with $1\ mole$ of a gas with $\gamma$ = $5/3$, then the value of $\gamma$ for the resulting mixture is
AIEEE 2002, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$C_v$ and $C_p$ denote the molar specific heat capacities of a gas at constant volume and constant pressure, respectively. ThenView Solution
- 2Ideal monoatomic gas is taken through a process $dQ = 2dU.$ The molar heat capacity for the processView Solution
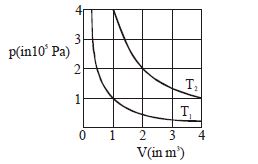
- 3The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution
- 4According to $C.E.$ van der Waal, the interatomic potential varies with the average interatomic distance $(R)$ asView Solution
- 5Which statements are correct about degrees of freedom?View Solution
$A.$ $A$ molecule with $n$ degrees of freedom has $n^{2}$ different ways of storing energy.
$B.$ Each degree of freedom is associated with $\frac{1}{2} RT$ average energy per mole.
$C.$ $A$ monoatomic gas molecule has $1$ rotational degree of freedom where as diatomic molecule has $2$ rotational degrees of freedom
$D$ $CH _{4}$ has a total to $6$ degrees of freedom Choose the correct answer from the option given below:
- 6The temperature of a gas at pressure $P$ and volume $V$ is $27°C.$ Keeping its volume constant if its temperature is raised to $927°C,$ then its pressure will beView Solution
- 7The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
- 8In gases of diatomic molecules, the ratio of the two specific heats of gases ${C_P}/{C_V}$ isView Solution
- 9$r.m.s.$ velocity of nitrogen molecules at $NTP$ is ...... $m/s$View Solution
- 10View SolutionInternal energy of a non-ideal gas depends on ..........
