Which statements are correct about degrees of freedom?
$A.$ $A$ molecule with $n$ degrees of freedom has $n^{2}$ different ways of storing energy.
$B.$ Each degree of freedom is associated with $\frac{1}{2} RT$ average energy per mole.
$C.$ $A$ monoatomic gas molecule has $1$ rotational degree of freedom where as diatomic molecule has $2$ rotational degrees of freedom
$D$ $CH _{4}$ has a total to $6$ degrees of freedom Choose the correct answer from the option given below:
JEE MAIN 2022, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 2$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will beView Solution
- 3At $300\,K$, the rms speed of oxygen molecules is $\sqrt{\frac{\alpha+5}{\alpha}}$ times to that of its average speed in the gas. Then, the value of $\alpha$ will be (used $\pi=\frac{22}{7}$ )View Solution
- 4At ..... $K$ temperature average speed of $O_2$ will be ${\left( {\frac{7}{4}} \right)^{th}}$ of rms speed of $N_2$ at $47\,^oC$View Solution
- 5A gas is filled in a cylinder, its temperature is increased by $20\%$ on kelvin scale and volume is reduced by $10\%$ . How much percentage of the gas will leak out at constant pressure ...... $\%$View Solution
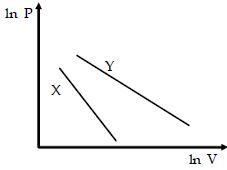
- 6For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
- 7$r.m.s.$ velocity of nitrogen molecules at $NTP$ is ...... $m/s$View Solution
- 8An ideal gas is initially at temperature $T$ and volume $V.$ Its volume is increased by $\Delta V$ due to an increase in temperature $\Delta T,$ pressure remaining constant. The quantity $\delta = \Delta V/(V\Delta T)$ varies with temperature asView Solution
- 9If the collision frequency of hydrogen molecules in a closed chamber at $27^{\circ} \mathrm{C}$ is $\mathrm{Z}$, then the collision frequency of the same system at $127^{\circ} \mathrm{C}$ is :View Solution
- 10The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution