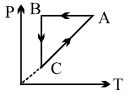
A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it would

Diffcult
In the given cyclic process, From $A$ to $B$, the pressure is constant.
From $C$ to $A, P \propto T$
Hence, the volume is constant.
From $B$ to $C$, the temperature is constant.
Hence, $P \propto \frac{1}{V}$
From $C$ to $A,$ volume is constant.
Hence, when presented on $P V,$ it would be represented by option $\mathrm{C}$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
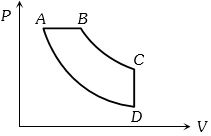
- 1View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 2In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 3In an adiabatic change, the pressure $P$ and temperature $T$ of a monoatomic gas are related by the relation $P \propto {T^C}$, where $C$ equalsView Solution
- 4During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
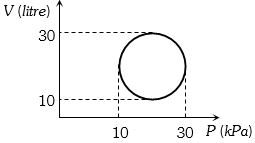
- 5View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
- 6Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
- 7A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
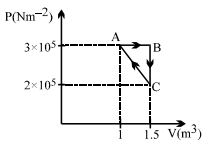
- 8Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
- 9Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
- 10Three samples of the same gas $A, B$ and $C(\gamma = 3/2)$ have initially equal volume. Now the volume of each sample is doubled. The process is adiabatic for $A$ isobaric for $B $ and isothermal for $C$. If the final pressures are equal for all three samples, the ratio of their initial pressures areView Solution
