In an adiabatic change, the pressure $P$ and temperature $T$ of a monoatomic gas are related by the relation $P \propto {T^C}$, where $C$ equals
AIIMS 2001,AIPMT 1994,AIIMS 2007, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 2An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 3The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
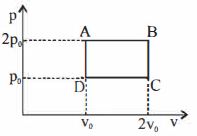
- 4A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$View Solution
- 5Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
- 6View SolutionIn the following figures heat is absorbed by the gas
- 7$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution
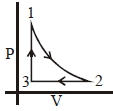
- 8Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 9A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 10The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:View Solution
