A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)
Diffcult
(b) $\Delta Q = mc\Delta T$ $⇒$ $\Delta T = \frac{{20000J}}{{1kg \times (400J/kg^\circ C)}} = 50^\circ C$
$⇒$ $T$ Final $= 70°C$
Hence $W = {P_{atm}}\Delta V = {P_{atm}}{V_0}\gamma \,\Delta T$
$ = ({10^5}N/{m^2})\,\left( {\frac{1}{{9 \times {{10}^3}}}{m^3}} \right)\,(9 \times {10^{ - 5}}/^\circ C)\,(50^\circ C) = 0.05J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 2View SolutionThe heat absorbed by a system in going through the given cyclic process is:
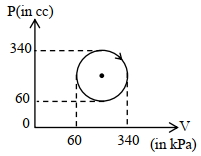
- 3View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
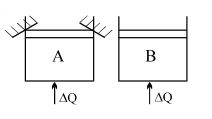
- 4Two identical vessels $A \& B$ contain equal amount of ideal monoatomic gas. The piston of $A$ is fixed but that of $B$ is free. Same amount of heat is absorbed by$A \& B$. If $B'$s internal energy increases by $100 \,\,J$ the change in internal energy of $A$ is ...... .$J$View Solution
- 5The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 6View SolutionWhich is incorrect
- 7View SolutionA Camot cycle consists of
- 8Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 9Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 10View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
