The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$
JEE MAIN 2021, Diffcult
We know that work done is
$W =\int PdV\quad \ldots (1)$
$\Rightarrow P=\frac{ nRT }{ V }\quad \ldots (2)$
$\Rightarrow W =\int \frac{ nRT }{ V } dv\quad \ldots (3)$
and $V = KT ^{2 . / 3}\quad \ldots (4)$
$\Rightarrow W =\int \frac{ nRT }{ KT ^{2 / 3}} \cdot dv \quad \ldots (5)$
$\Rightarrow$ from (4)$: dv =\frac{2}{3} KT ^{-1 / 3} dT$
$\Rightarrow W =\int_{ T _{1}}^{ T _{2}} \frac{ nRT }{ KT ^{2 / 3}} \frac{2}{3} K \frac{1}{ T ^{1 / 3}} dT$
$\Rightarrow W =\frac{2}{3} n R \times\left( T _{2}- T _{1}\right) \ldots(6)$
$\Rightarrow T _{2}- T _{1}=90 K \ldots(7)$
$\Rightarrow W=\frac{2}{3} n R \times 90$
$\Rightarrow W =60 nR$
Assuming $1$ mole of gas
$n =1$
So $W =60 \,R$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
- 2View SolutionIn an isothermal change, an ideal gas obeys
- 3A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 4View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 5For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
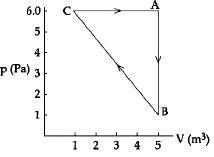
- 6View SolutionIn which thermodynamic process, volume remains same
- 7A Carnot engine has an efficiency of $50 \%$ when its source is at a temperature $327^{\circ}\,C$. The temperature of the sink is $.........^{\circ} C$View Solution
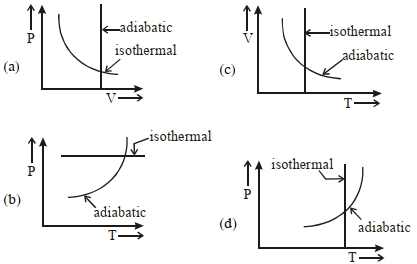
- 8View SolutionWhich one is the correct option for the two different thermodynamic processes ?
- 9View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 10A sample of an ideal gas is taken through a cycle a shown in figure. It absorbs $50J$ of energy during the process $AB$, no heat during $BC$, rejects $70J$ during $CA.$ $40J$ of work is done on the gas during $BC$. Internal energy of gas at $A$ is $1500J$, the internal energy at $C$ would be ........ $J$View Solution