Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
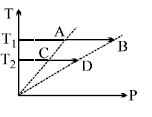
- 2On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
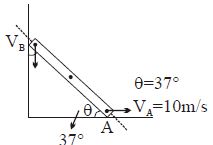
- 3Find $V_B = ?$View Solution
- 4A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 5A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 6$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 7The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 8Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 9View SolutionIn an isothermal change, an ideal gas obeys
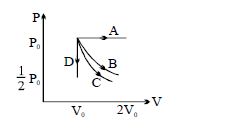
- 10The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution