One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)
Medium
$\Delta \mathrm{U}=-\mathrm{W}=\frac{\mathrm{nR}\left(\mathrm{T}_{1}-\mathrm{T}_{2}\right)}{1-\mathrm{r}}=\frac{1 \times 8.3(-8)}{1-1.4}=166 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 2An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 3The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 4A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
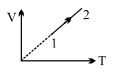
- 5An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
- 6One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
- 7A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$View Solution
- 8Given below are two statements:View Solution
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
- 9A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 10A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
