A gas molecule of mass $M$ at the surface of the Earth has kinetic energy equivalent to $0\,^oC$. If it were to go up straight without colliding with any other molecules, how high it would rise? Assume that the height attained is much less than radius of the earth. ($k_B$ is Boltzmann constant)
JEE MAIN 2014, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
- 2The root mean square speed of hydrogen molecules at $300$ $K$ is $1930\, m/s.$ Then the root mean square speed of oxygen molecules at $900\, K$ will be ....... $m/s$View Solution
- 3$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 4A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
- 5The volume of a gas at $21°C$ temperature and $768 \,mm$ pressure is $1\, litre$. If the density of the gas is $1.2\, gm/litre$ at $NTP,$ then its mass will be ..... $gm$View Solution
- 6A flask contains hydrogen and oxygen in the ratio of $2: 1$ by mass at temperature $27^{\circ} C$. The ratio of average kinetic energy per molecule of hydrogen and oxygen respectively is :View Solution
- 7When temperature of an ideal gas is increased from $27^\circ C$ to $227^\circ C$, its $r.m.s.$ speed changed from $400\, metre/sec$ to ${V_s}.$ The ${V_s}$ is ........ $metre/sec$View Solution
- 8For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
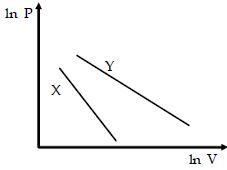
- 9View SolutionIf two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats is
- 10Energy of all molecules of a monoatomic gas having a volume $V$ and pressure $P$ is $\frac{3}{2}PV$. The total translational kinetic energy of all molecules of a diatomic gas as the same volume and pressure isView Solution
