$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?
JEE MAIN 2017, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A spherical soap bubble inside an air chamber at pressure $P_0=10^5 Pa$ has a certain radius so that the excess pressure inside the bubble is $\Delta P=144 Pa$. Now, the chamber pressure is reduced to $8 P_0 / 27$ so that the bubble radius and its excess pressure change. In this process, all the temperatures remain unchanged. Assume air to be an ideal gas and the excess pressure $\Delta P$ in both the cases to be much smaller than the chamber pressure. The new excess pressure $\Delta P$ in $Pa$ isView Solution
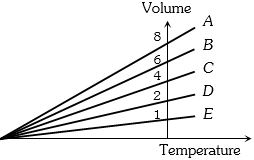
- 2The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 3The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at $0°C$ is $3180$ metres/second. The pressure on the hydrogen gas is ..... $atm$ (Density of hydrogen gas is $8.99 \times {10^{ - 2}}\,kg/{m^3}$, $1$ atmosphere $ = 1.01 \times {10^5}\,N/{m^2})$View Solution
- 4A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 5View SolutionKinetic theory of gases provide a base for
- 6View SolutionVolume of gas become four times if
- 7A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
- 8A vessel contains $16 \,g$ of hydrogen and $128 \,g$ of oxygen at standard temperature and pressure. The volume of the vessel in $cm ^{3}$ isView Solution
- 9If the root mean square velocity of the molecules of hydrogen at $NTP$ is $1.84\, km/s$. Calculate the root mean square velocity of oxygen molecule at $NTP$, molecular weight of hydrogen and oxygen are $2$ and $32$ respectively ....... $km/sec$View Solution
- 10The ratio of total energy of all molecules of one mole $O_2$ to the total energy of all molecules of two moles of $He$ at the same temprature isView Solution
