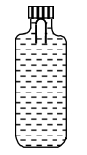
A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?

Medium
for cyclic process
$\mathrm{dQ}=\mathrm{d} \mathrm{W}$
$\Rightarrow \mathrm{d} \mathrm{Q}=\frac{1}{2} \times 200 \times 100 \times 10^{-6} \times 10^{3} \mathrm{J}=10 \mathrm{Joule}$
also $dQ$ $=2.4 \mathrm{cal}$
$\Rightarrow \mathrm{J}=\frac{10}{2.4}=4.17 \mathrm{J} / \mathrm{cal}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 2View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 3During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
- 4A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
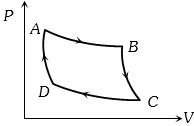
- 5Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
- 6Two cylinders $A$ and $B$ of equal capacity are connected to each other via a stop cock. A contains an Ideal gas at standard temperature and pressure. $B$ is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :View Solution
- 7A vertical cylinder with heat-conducting walls is closed at the bottom and is fitted with a smooth light piston. It contains one mole of an ideal gas. The temperature of the gas is always equal to the surrounding’s temperature, $T_0$. The piston is moved up slowly to increase the volume of the gas to $\eta$ times. Which of the following is incorrect?View Solution
- 8If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
- 9Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 10A soft plastic bottle, filled with water of density $1 gm / cc$, carries an inverted glass test-tube with some air (ideal gas) trapped as shown in the figure. The test-tube has a mass of $5 gm$, and it is made of a thick glass of density $2.5 gm / cc$. Initially the bottle is sealed at atmospheric pressure $p_0=10^5 Pa$ so that the volume of the trapped air is $v_0=3.3 cc$. When the bottle is squeezed from outside at constant temperature, the pressure inside rises and the volume of the trapped air reduces. It is found that the test tube begins to sink at pressure $P_0+\Delta p$ without changing its orientation. At this pressure, the volume of the trapped air is $v_0-\Delta v$.View Solution
Let $\Delta v=X$ cc and $\Delta p=Y \times 10^3 Pa$.
($1$) The value of $X$ is
($2$) The value of $Y$ is
Give the answer or quetion ($1$) and ($2$)