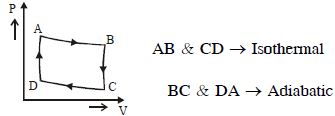Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statements
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
Diffcult
The area under the $P-V$ curve gives the work done by the gas.
since initial and final conditions are the same, hence internal energy has not changed which is
a function of $T$ that is temperature. $\Delta U=0$
$Q=W+\Delta U=W$
Hence, the area also shows heat absorbed as it is equal to work done by hte gas.
Answer- $(I I, I I I)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
- 2Two cylinders $A$ and $B$ of equal capacity are connected to each other via a stop cock. A contains an Ideal gas at standard temperature and pressure. $B$ is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :View Solution
- 3View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 4$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
- 5When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 6An ideal monoatomic gas with pressure $P$, volume $V$ and temperature $T$ is expanded isothermally to a volume $2\, V$ and a final pressure $P_i$. If the same gas is expanded adiabatically to a volume $2\,V$, the final pressure is $P_a$ . The ratio $\frac{{{P_a}}}{{{P_i}}}$ isView Solution
- 7A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 8If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 9One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
- 10The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution