A piece of iron is heated in a flame. It first becomes dull red then becomes reddish yellow and finally turns to white hot. The correct explanation for the above observation is possible by using
AIPMT 2013, Easy
According to $Wien's$ displacement law
${\lambda _m}T = constant$
${\lambda _m} = \frac{{constant}}{T}$
So when a piece of iron is heated, ${\lambda _m}$ decreases $i.e.,$ with rise in temperature the maximum intensity of radiation emitted gets shifted towards the shorter wavelengths. So the colour of the heated object will change that of longer wavelength $(red)$ to that of shorter $(reddish\,yellow)$ and when the temperature is sufficiently high and all wavelengths are emitted, the colour will become white.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn steady state graph between temp and distance from hot end is
- 2If temperature of a black body increases from ${7^o}C$ to ${287^o}C$, then the rate of energy radiation increases byView Solution
- 3Two identical rods of copper and iron are coated with wax uniformly. When one end of each is kept at temperature of boiling water, the length upto which wax melts are $8.4cm$ and $4.2cm$ respectively. If thermal conductivity of copper is $0.92$ , then thermal conductivity of iron isView Solution
- 4The energy distribution $E$ with the wavelength $(\lambda )$ for the black body radiation at temperature $T\;Kelvin$is shown in the figure. As the temperature is increased the maxima willView Solution
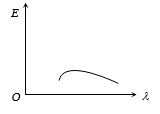
- 5Two conducting rods $A$ and $B$ of same length and cross-sectional area are connected $(i)$ In series $(ii)$ In parallel as shown. In both combination a temperature difference of $100^o C$ is maintained. If thermal conductivity of $A$ is $3K$ and that of $B$ is $K$ then the ratio of heat current flowing in parallel combination to that flowing in series combination isView Solution
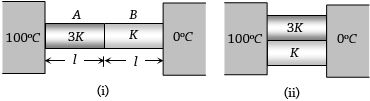
- 6A composite block is made of slabs $A, B, C, D$ and $E$ of different thermal conductivities (given in terms of a constant $K$ ) and sizes (given in terms of length, $L$ ) as shown in the figure. All slabs are of same width. Heat $'Q'$ flows only from left to right through the blocks. Then in steady state $Image$View Solution
$(A)$ heat flow through $A$ and $E$ slabs are same.
$(B)$ heat flow through slab $E$ is maximum.
$(C)$ temperature difference across slab $E$ is smallest.
$(D)$ heat flow through $C =$ heat flow through $B +$ heat flow through $D$.
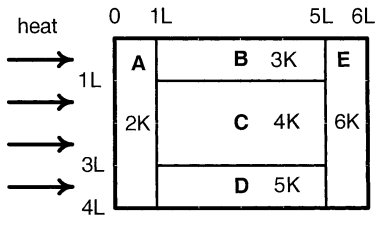
- 7Column $I$ gives some devices and Column $II$ gives some process on which the functioning of these devices depend. Match the devices in Column $I$ with the processes in Column $II$ and indicate your answer by darkening appropriate bubbles in the $4 \times 4$ matrix given in the $ORS$.View Solution
Column $I$ Column $II$ $(A)$ Bimetallic strip $(p)$ Radiation from a hot body $(B)$ Steam engine $(q)$ Energy conversion $(C)$ Incandescent lamp $(r)$ Melting $(D)$ Electric fuse $(s)$ Thermal expansion of solids - 8A black body has maximum wavelength ${\lambda _m}$ at temperature $2000\; K$. Its corresponding wavelength at temperature $3000 \;K$ will beView Solution
- 9Three very large plates of same area are kept parallel and close to each other. They are considered as ideal black surfaces and have very high thermal conductivity. The first and third plates are maintained at temperatures $2T$ and $3T$ respectively. The temperature of the middle (i.e. second) plate under steady state condition isView Solution
- 10View SolutionThe energy supply being cut-off, an electric heater element cools down to the temperature of its surroundings, but it will not cool further because
