$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with 8 reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^{\circ}\,C$ to final temperature $200^{\circ}\,C$. Entropy change of the body in the two cases respectively is :
Download our appand get started for free
Similar Questions
- 1A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 2View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 3A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
- 4A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
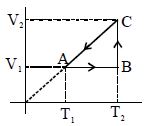
- 5View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
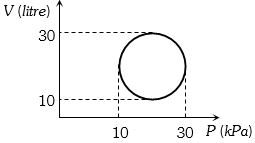
- 6View SolutionThe heat absorbed by a system in going through the given cyclic process is:
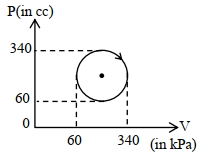
- 7Carbon monoxide is carried around a closed cycle $abc$ in which bc is an isothermal process as shown in the figure. The gas absorbs $7000 J$ of heat as its temperature increases from $300 K$ to $1000 K$ in going from $a$ to $b$. The quantity of heat rejected by the gas during the process $ca$ is ..... $J$View Solution
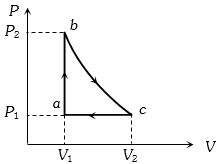
- 8Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 9View SolutionEntropy of a system decreases .........
- 10A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
