According to the kinetic theory of gases, at absolute temperature
AIIMS 1998,AIPMT 1990, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A closed vessel contains $10 g$ of an ideal gas $X$ at $300 \ K$, which exerts $2$ atm pressure. At the same temperature, $80 g$ of another ideal gas $Y$ is added to it and the pressure becomes $6 atm$. The ratio of root mean square velocities of $X$ and $Y$ at $300 \ K$ isView Solution
- 2Relation between $U, P$ and $V$ for ideal gas is $U= 2+2PV$ then gas isView Solution
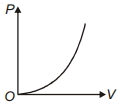
- 3The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 4View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
- 5A sample of gas is at $0°C.$ To what temperature it must be raised in order to double the $r.m.s.$ speed of the molecule ....... $^oC$View Solution
- 6A gas has $n$ degrees of freedom. The ratio of specific heat of gas at constant volume to the specific heat of gas at constant pressure will be.View Solution
- 7The diameter of oxygen molecule is $2.94 \times {10^{ - 10}}m.$ The Vander Waal’s gas constant ‘$b’$ in ${m^3}/mol$ will beView Solution
- 8An ideal gas of Molar mass $M$ is contained in a vertical tube of height $H$, closed at both ends. The tube is accelerating vertically upwards with acceleration $g$ Then, the ratio of pressure at the bottom and the mid point of the tube will beView Solution
- 9A polyatomic gas with $n$ degrees of freedom has a mean energy per molecule given byView Solution
($N$ is Avogadro's number)
- 10The temperature of argon, kept in a vessel, is raised by $1^\circ C$ at a constant volume. The total heat supplied to the gas is a combination of translational and rotational energies. Their respective shares areView Solution
