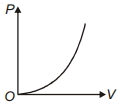
The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process is

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionYou have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
- 2$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
- 3Select the incorrect statement$(s)$View Solution
- 4View SolutionAt a given volume and temperature, the pressure of a gas
- 5The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 6$1$ mole of rigid diatomic gas performs a work of $Q / 5$ when heat $Q$ is supplied to it. The molar heat capacity of the gas during this transformation is $\frac{ x R }{8},$ The value of $x$ is $\ldots \ldots \ldots .$ $[ K =$ universal gas constant $]$View Solution
- 7An engineer is given a fixed volume $V_m$ of metal with which to construct a spherical pressure vessel. Interestingly, assuming the vessel has thin walls and is always pressurized to near its bursting point, the amount of gas the vessel can contain, $n$ (measured in moles), does not depend on the radius $r$ of the vessel : instead it depends only on $V_m$ (measured in $m^3$) the temperature $T$ (mensured in $K$). the ideal gas constant $R$ (measured in $J/(K\ mol$ )), and the tensile strength of the metal $\sigma $ (measured in $N/m^2$ ) . Which of the following gives $n$ in terms of these parameters?View Solution
- 8Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
- 9A container with insulating walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure $P$ and temperature $T$, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will beView Solution
- 10If the molecular weight of two gases are $M_1$ and $ M_1$, then at a temperature the ratio of root mean square velocity $v_1$ and $v_2$ will beView Solution
