Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$
Medium
(b)$\frac{{{\rm{Adiabatic elasticicity}}\;({E_\varphi })}}{{{\rm{Isothermal}}\;{\rm{elasticicity}}\;({E_\theta })}} = \gamma $
==>${E_\theta } = \frac{{{E_\varphi }}}{\gamma }$
==> ${E_\theta } = \frac{{2.1 \times {{10}^5}}}{{1.4}}$$ = 1.5 \times {10^5}N/{m^2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
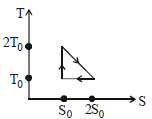
- 2View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 3For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 4An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
- 5A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
- 6View SolutionCan two isothermal curves cut each other
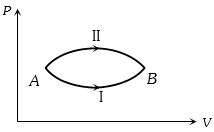
- 7A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
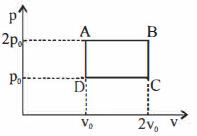
- 8The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
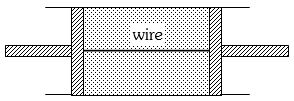
- 9A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
- 10A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
