A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$
JEE MAIN 2023, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a heat engine, the temperature of the source and sink are $500\, K$ and $375\, K$. If the engine consumes $25\times10^5\, J$ per cycle, the work done per cycle isView Solution
- 2A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 3Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
- 4If $300 ml$ of a gas at ${27^o}C$ is cooled to ${7^o}C$ at constant pressure, then its final volume will be ...... $ml$View Solution
- 5View SolutionIn the above thermodynamic process, the correct statement is
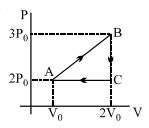
- 6A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
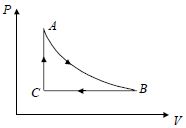
- 7A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
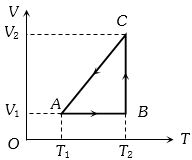
- 8$1\,kg$ of water at $100\, ^{\circ}C$ is converted into steam at $100^{\circ}\,C$ by boiling at atmospheric pressure. The volume of water changes from $1.00 \times 10^{-3}\,m ^3$ as a liquid to $1.671\,m ^3$ as steam. The change in internal energy of the system during the process will be $........kJ$ (Given latent heat of vaporisaiton $=2257\,kJ / kg$. Atmospheric pressure $=1 \times 10^5\,Pa$ )View Solution
- 9$Assertion :$ Reversible systems are difficult to find in real world.View Solution
$Reason :$ Most processes are dissipative in nature. - 10A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
