Air in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
AIIMS 2000, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
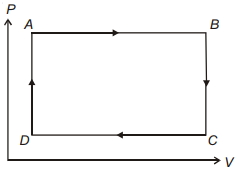
- 1The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
- 2During an adiabatic expansion of $2\, moles$ of a gas, the change in internal energy was found $-50J.$ The work done during the process is ...... $J$View Solution
- 3One mole of an ideal gas at an initial temperature of $T\, K$ does $6\, R\, joules$ of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $\frac{5}{3}$ , the final temperature of gas will beView Solution
- 4A Carnot engine with efficiency $50\,\%$ takes heat from a source at $600\,K$. In order to increase the efficiency to $70\,\%$, keeping the temperature of sink same, the new temperature of the source will be $.........\,K$View Solution
- 5View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
- 6Two moles of monoatomic gas is expanded from $(P_0, V_0)$ to $(P_0 , 2V_0)$ under isobaric condition. Let $\Delta Q_1$, be the heat given to the gas, $\Delta W_1$ the work done by the gas and $\Delta U_1$ the change in internal energy. Now the monoatomic gas is replaced by a diatomic gas. Other conditions remaining the same. The corresponding values in this case are $\Delta Q_2 , \Delta W_2 , \Delta U_2$ respectively, thenView Solution
- 7A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
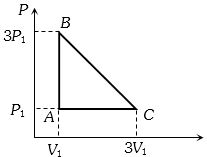
- 8An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
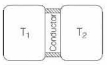
- 9A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
- 10Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
