==> $\Delta W = - ( - 50) = + 50J$
Download our appand get started for free
Similar Questions
- 1The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 2Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
- 3View SolutionWhich one is the correct option for the two different thermodynamic processes ?
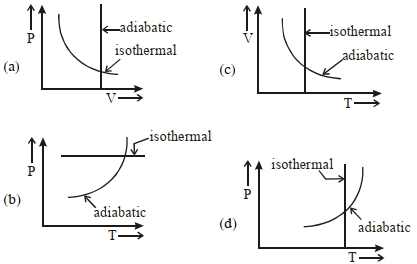
- 4View SolutionIn the above thermodynamic process, the correct statement is
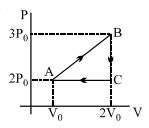
- 5$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):View Solution
- 6Answer the following by appropriately matching the lists based on the information given in the paragraph.View Solution
In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
List-$I$ List-$II$ $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ $(P)$ $\frac{1}{3} R T_0 \ln 2$ $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ $(Q)$ $\frac{1}{3} RT _0$ $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ $(R)$ $R T _0$ $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ $(S)$ $\frac{4}{3} RT _0$ $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ $(U)$ $\frac{5}{6} RT _0$ If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$
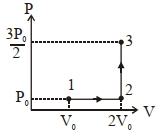
- 7A cyclic process is shown in figure. Work done during isobaric expansion is ...... $J$View Solution
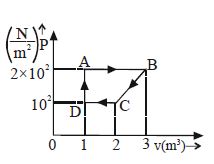
- 8View SolutionWhich statement is incorrect?
- 9View SolutionFor an isometric process
- 10An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
