An alkene $\ce{CH_3CH=CH_2}$ is treated with $\ce{B_2H_6}$ in presence of $\ce{H_2O_2}.$ The final product formed is$:$
Hydroboration$-$oxidation reaction follows anti-Markovnikov's addition of $\ce{H−OH}$ across $\ce{C=C}$ to give alcohol.
Thus an alkene $\ce{CH_3CH=CH2}$ when treated with $\ce{B_2H_{6}}$ in presence of $\ce{H_2O_2}$ will yield the final product as $\ce{CH_3CH_2CH_{2}OH}$
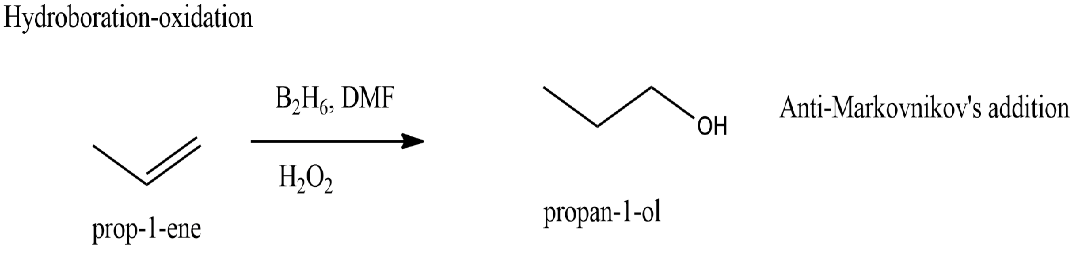
Thus an alkene $\ce{CH_3CH=CH2}$ when treated with $\ce{B_2H_{6}}$ in presence of $\ce{H_2O_2}$ will yield the final product as $\ce{CH_3CH_2CH_{2}OH}$

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$\ce{IUPAC}$ name of $m-$cresol is $.......$View Solution
- 2View SolutionWhich of the following phenols has lowest solubility in water?
- 3The $\text{IUPAC}$ name of $\ce{CH_{3}−C(CH_{3})(OH)CH_2−CH(CH_3)_2}$ is:View Solution
- 4View SolutionAlcohols containing only up to ________________ carbon atoms are completely miscible with water.
- 5View SolutionAn ester can be prepared by the reaction of:
- 6View SolutionWhich of the following alcohols is the least soluble in water?
- 7In iodine charge transfer test the color of ether compound formed is$:$View Solution
- 8View SolutionWhich of the following combinations can be used for the preparation of cis vic diol?
- 9View SolutionThe Product formed by the reaction of phenol with Zn dust
- 10$3-$methyl$-2-$pentene on reaction with $\ce{HOCl}$ gives$:$View Solution
