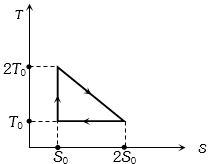
An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle is

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 2View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
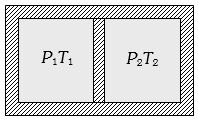
- 3Following figure shows on adiabatic cylindrical container of volume ${V_0}$ divided by an adiabatic smooth piston (area of cross-section = $A$ ) in two equal parts. An ideal gas $({C_P}/{C_V} = \gamma )$ is at pressure $P_1$ and temperature $T_1$ in left part and gas at pressure $P_2$ and temperature $T_2$ in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose $ x$ = displacement of the piston)View Solution
- 4$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
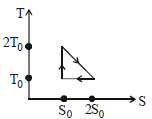
- 5View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 6Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
- 7A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
- 8An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 9View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 10View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is