Match List$-I$ with List$-II$
| List$-I$ | List$-II$ |
| $(a)$ Isothermal | $(i)$ Pressure constant |
| $(b)$ Isochoric | $(ii)$ Temperature constant |
| $(c)$ Adiabatic | $(iii)$ Volume constant |
| $(d)$ Isobaric | $(iv)$ Heat content is constant |
Choose the correct answer from the options given below
JEE MAIN 2021, Medium
$(a)$ Isothermal $\Rightarrow$ Temperature constant
$(a)$ $\rightarrow$ $(ii)$
$(b)$ Isochoric $\Rightarrow$ Volume constant
$(a)$ $\rightarrow$ $(iii)$
$(c)$ Adiabatic $\Rightarrow \Delta Q=0$
$\Rightarrow$ Heat content is constant
$(c)$ $\rightarrow$ $(iv)$
$(d)$ Isobaric $\Rightarrow$ Pressure constant
$(d)$ $\rightarrow$ $(i)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 2$\Delta U + \Delta W = 0$ is valid forView Solution
- 3A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
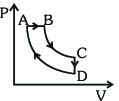
- 4An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 5$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 6Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 7The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$View Solution
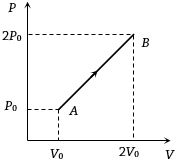
- 8A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
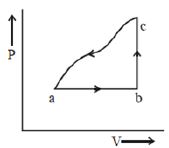
- 9View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
- 10View SolutionWhich of the following statements is correct for any thermodynamic system
