An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :
JEE MAIN 2016, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
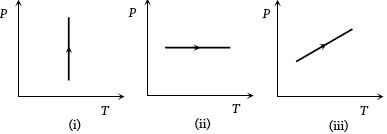
- 1View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
- 2A gas at ${27}^\circ C$ temperature and $30$ atmospheric pressure is allowed to expand to the atmospheric pressure. If the volume becomes $10$ times its initial volume, then the final temperature becomes ...... $^oC$View Solution
- 3A mixture of two moles of hydrogen and one mole of argon gas is taken in a closed container at room temperature. Consider the following two statementsView Solution
$(i)$ The average kinetic energy of each molecule of $H_2$ and $Ar$ are the same.
$(ii)$ The partial pressure due to argon gas is more than that due to hydrogen gas
- 4View SolutionA piston is slowly pushed into a metal cylinder containing an ideal gas. Which of the following statements is/are incorrect?
- 5View SolutionSelect the appropriate property of an ideal gas
- 6The $r.m.s.$ speed of the molecules of a gas at a pressure ${10^5}$ $Pa$ and temperature $0^\circ C$ is $0.5km\;{\sec ^{ - 1}}.$ If the pressure is kept constant but temperature is raised to $819°C,$ the velocity will become ........ $km\,sec^{-1}$View Solution
- 7The temperature of the mixture of one mole of helium and one mole of hydrogen is increased from ${0^o}C$ to ${100^o}C$ at constant pressure. The amount of heat delivered will be ...... $cal$View Solution
- 8The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
- 9The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
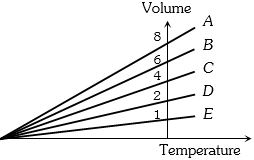
- 10Two ideal polyatomic gases at temperatures $T _{1}$ and $T _{2}$ are mixed so that there is no loss of energy. If $F _{1}$ and $F _{2}, m _{1}$ and $m _{2}, n _{1}$ and $n _{2}$ be the degrees of freedom, masses, number of molecules of the first and second gas respectively, the temperature of mixture of these two gases isView Solution
