The temperature of the mixture of one mole of helium and one mole of hydrogen is increased from ${0^o}C$ to ${100^o}C$ at constant pressure. The amount of heat delivered will be ...... $cal$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A jar contains a gas and few drops of water at $T K.$ The pressure in the jar is $830 \,mm$ of mercury. The temperature of jar is reduced by $1\%.$ The saturated vapour pressure of water at the two temperatures are $30 \,mm$ and $ 25\, mm$ of mercury. Then the new pressure in the jar will be ..... $mm$ of $Hg$View Solution
- 2The temperature at which the average translational kinetic energy of a molecule is equal to the energy gained by an electron in accelerating from rest through a potential difference of $1\, volt$ isView Solution
- 3The specific heat of a gas at constant volume is $21.2\, J/mole/°C.$ If the temperature is increased by $1°C$ keeping the volume constant, the change in its internal energy will be ...... $J$View Solution
- 4The specific heats, $C_P$ and $C_V$ of a gas of diatomic molecules, $A$, are given (in units of $J\, mol^{-1}\, K^{-1}$) by $29$ and $22$, respectively. Another gas of diatomic molecules $B$, has the corresponding values $30$ and $21$. If they are treated as ideal gases, thenView Solution
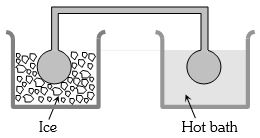
- 5Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution
- 6View SolutionIn kinetic theory of gases, which of the following statements regarding elastic collisions of the molecules is wrong
- 7Two ideal polyatomic gases at temperatures $T _{1}$ and $T _{2}$ are mixed so that there is no loss of energy. If $F _{1}$ and $F _{2}, m _{1}$ and $m _{2}, n _{1}$ and $n _{2}$ be the degrees of freedom, masses, number of molecules of the first and second gas respectively, the temperature of mixture of these two gases isView Solution
- 8On any planet, the presence of atmosphere implies (${C_{rms}}$= root mean square velocity of molecules and ${V_e}$= escape velocity)View Solution
- 9View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 10The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
