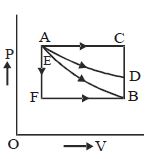
An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum in

Medium
$\Delta Q = \Delta W + dU$ for all path $dU$ will be same due to state function. and work done will be minimum in $AFB$ path so energy given to system is min in $AFB$ path.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA measure of the degree of disorder of a system is known as
- 2Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
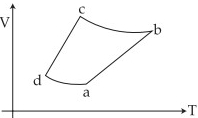
- 3An ideal gas goes through a reversible cycle $a\to b\to c\to d$ has the $V - T$ diagram shown below. Process $d\to a$ and $b\to c$ are adiabatic.... The corresponding $P - V$ diagram for the process is (all figures are schematic and not drawn to scale)View Solution
- 4A diatomic ideal gas is compressed adiabatically to $\frac{1}{32}$ of its initial volume. If the initial temperature of the gas is $T_1$ (in Kelvin) and the final temperature is $a T_1$, the value of $a$ isView Solution
- 5If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 6The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process(es) does the temperature of the gas decrease ?View Solution
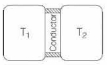
- 7A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
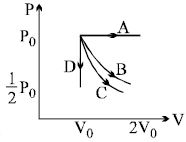
- 8The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
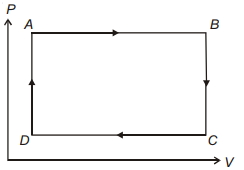
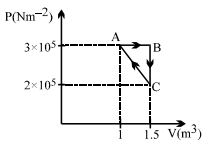
- 9The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
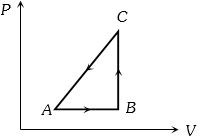
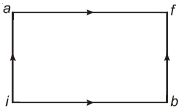
- 10When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution