$\Delta S=\frac{Q}{T}$
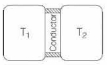
Now, for given system to be in steady state, heat lost by resorvoir at temperature $T_{1}=$ heat gained by resorvoir at temperature $T_{2}$ (=Q say).
So, change in entropy for heat conduction process is
$\Delta S-\frac{-Q}{}+\frac{(+Q)}{T_{1}}$
$\Rightarrow \quad \Delta S=Q\left(\frac{1}{T_{2}}-\frac{1}{T_{1}}\right)$
So, time rate of change of entropy is
$\frac{d S}{d t}=\frac{d}{d t}\left\{Q\left(\frac{1}{T_{2}}-\frac{1}{T_{1}}\right)\right\}$
$=\left(\frac{1}{T_{2}}-\frac{1}{T_{1}}\right) \cdot\left(\frac{d Q}{d t}\right)$
$\frac{d S}{d t}=\left(\frac{T_{1}}{T_{2}}-1\right) \cdot T_{1}\left(\frac{d Q}{d t}\right)$
As, $\quad \frac{d Q}{d t}=-k A\left(\frac{d T}{d x}\right)$
So, $\quad \frac{d S}{d t}=-k A \frac{d T}{d x} \cdot T_{1}\left(\frac{T_{1}}{T_{2}}-1\right)$
$\Rightarrow \quad \frac{d S}{d t}=k A T_{1} \cdot\left(1-\frac{T_{1}}{T_{2}}\right) \cdot \frac{d T}{d x}$
$=\frac{k A T_{1}}{x}\left(1-\frac{T_{1}}{T_{2}}\right)\left(T_{1}-T_{2}\right)$
$=\frac{k A T_{1}^{2}}{x}\left(1-\frac{T_{1}}{T_{2}}\right)\left(1-\frac{T_{2}}{T_{1}}\right)$
Clearly, at $\frac{T_{1}}{T_{2}}=1, \frac{d S}{d t}=0$.
Also, graph is asym metrical. So, correct option is $(b)$.
Download our appand get started for free
Similar Questions
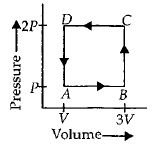
- 1A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
- 2A Carnot engine whose heat $\operatorname{sinks}$ at $27\,^{\circ} C$, has an efficiency of $25 \%$. By how many degrees should the temperature of the source be changed to increase the efficiency by $100 \%$ of the original efficiency $?$View Solution
- 3For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 4A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
- 5A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 6View SolutionFirst law thermodynamics states that
- 7Two moles of monoatomic gas is expanded from $(P_0, V_0)$ to $(P_0 , 2V_0)$ under isobaric condition. Let $\Delta Q_1$, be the heat given to the gas, $\Delta W_1$ the work done by the gas and $\Delta U_1$ the change in internal energy. Now the monoatomic gas is replaced by a diatomic gas. Other conditions remaining the same. The corresponding values in this case are $\Delta Q_2 , \Delta W_2 , \Delta U_2$ respectively, thenView Solution
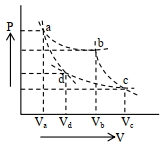
- 8Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution
- 9$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
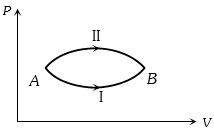
- 10A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution