At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$
Medium
(c)${T_2} = {T_1}{\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} = 273{(2)^{0.41}}$$ = 273 \times 1.328 = 363K$
$W = \frac{{R({T_1} - {T_2})}}{{\gamma - 1}} = \frac{{8.31(273 - 363)}}{{1.41 - 1}}$$ = - \,1824$
==> $|W| \approx 1815 J$
$W = \frac{{R({T_1} - {T_2})}}{{\gamma - 1}} = \frac{{8.31(273 - 363)}}{{1.41 - 1}}$$ = - \,1824$
==> $|W| \approx 1815 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 2A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
- 3A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
- 4Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$View Solution
- 5Work done for the process shown in the figure is ............ $J$View Solution
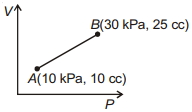
- 6A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 7A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
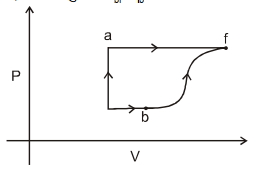
- 8Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
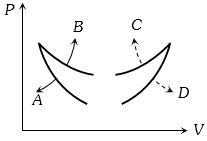
- 9During an experiment, an ideal gas is found to obey a condition $VP^2 =$ constant. The gas is initially at a temperature $T$, pressure $P$ and volume $V.$ The gas expands to volume $4V$.View Solution
- 10A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
