Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in addition
KVPY 2011, Diffcult
(b)
Compression of a gas in a compressor is nearly an adiabatic process.
So, by using $p_{\text {in }}^{1-\gamma} \cdot T_{\text {in }}^\gamma=p_{\text {out }}^{1-\gamma} \cdot T_{\text {out }}^\gamma$
We get, $(0.28)^{1-\gamma}(233)^\gamma=(1)^{1-\gamma}(T)^\gamma$
Here, for air, $\gamma=14=\frac{7}{5}$
Hence, $T=(233)(0.28)^{-2 / 7}=\frac{233}{(0.28)^{2 / 7}}$
This temperature is much higher than $298 \,K$ or $25^{\circ} C$.
So, an air-conditioner is needed to cool the air coming out of compressor.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn thermodynamic processes which of the following statements is not true?
- 2View SolutionThe first law of thermodynamics is concerned with the conservation of
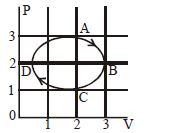
- 3The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
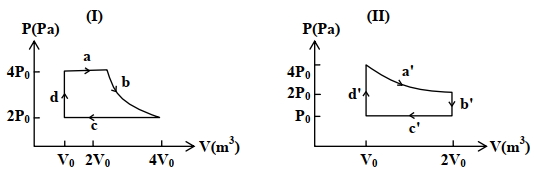
- 4One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the $P-V$ diagrams below. In cycle I, processes $a, b, c$ and $d$ are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes $a^{\prime}, b^{\prime}, c^{\prime}$ and $d^{\prime}$ are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is $W_I$ and that during cycle II is $W_{I I}$. The ratio $W_I / W_{I I}$ is . . . .View Solution
(image)
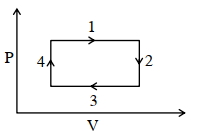
- 5An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
- 6View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 7View SolutionIn which thermodynamic process, volume remains same
- 8If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 9Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 10Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
