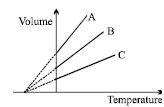
The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2\, m$ at a pressure $2\,P$ is given by the straight line

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.View Solution
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
- 2$1$ mole of gas occupies a volume of $100\, ml$ at $50\, mm$ pressure. What is the volume occupied by two moles of gas at $100\, mm$ pressure and at same temperature ..... $ml$View Solution
- 3An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
- 4Sound travels in a mixture of two moles of helium and n moles of hydrogen. If rms speed of gas molecules in the mixture is $\sqrt{2}$ times the speed of sound, then the value of $n$ will beView Solution
- 5The kinetic energy of translation of $20\, gm$ of oxygen at $47°C$ is (molecular wt. of oxygen is $32 \,gm/mol$ and $R = 8.3\, J/mol/K)$View Solution
- 6$Assertion :$ For a gas atom the number of degrees of freedom is $3$.View Solution
$Reason :$ $\frac{{{C_P}}}{{{C_V}}} = \gamma $ - 7The average translational energy and the $r.m.s.$ speed of molecules in a sample of oxygen gas at $300 K$ are $6.21 \times {10^{ - 21}}\,J$ and $484\, m/s$ respectively. The corresponding values at $600 K$ are nearly (assuming ideal gas behaviour)View Solution
- 8At ...... $ ^o C$ temperature, the mean kinetic energy of ${O_2}$ will be the same for ${H_2}$ molecules at $- 73^o C$View Solution
- 9Gas at a pressure ${P_0}$ in contained is a vessel. If the masses of all the molecules are halved and their speeds are doubled, the resulting pressure $P$ will be equal toView Solution
- 10A gas mixture consists of $2$ moles of $O_2$ and $4$ moles of $Ar$ at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
