Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
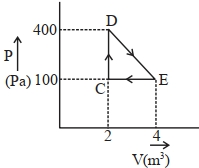
- 1A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
- 2View SolutionWhich of the following can be coefficient of performance of refrigerator?
- 3A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 4A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 5If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution
- 6$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
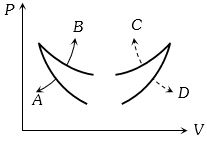
$Reason :$ Dissipative effects cannot be eliminated. - 7Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
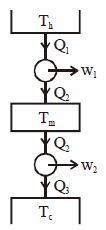
- 8Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
- 9View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 10View SolutionFirst law thermodynamics states that
