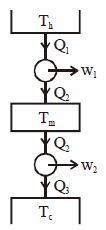
Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given by

Diffcult
$ \eta_{net} $$=\frac{\omega_1+\omega_2}{\theta_1}$
$ \eta_1=$$ \frac{w_1}{\theta_1} \eta_2=\frac{w_2}{\theta_2} $
$ \eta_{\eta_{e t}}$$=\frac{\eta_1 q_1+\eta_2 q_2}{\theta_1} $
Now, $=1-\frac{\theta_2}{\theta_1} $
$ \frac{\theta_2}{\theta_1} $$=1-\eta_1 $
$ \eta_{net} $$=n_1+\left(1-\eta_1\right) \eta_2 $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
- 2View SolutionIn Carnot engine the work done by working substance is equivalent to
- 3View SolutionCofficient of performance of refigerator is
- 4Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 5An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 6View SolutionA measure of the degree of disorder of a system is known as
- 7$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 8A Carnot engine, having an efficiency of $\eta = 1/10$ as heat engine, is used as a refrigerator. If the work done on the system is $10\ J$, the amount of energy absorbed from the reservoir at lower temperature is ....... $J$View Solution
- 9$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 10Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
