$\frac{1}{2} $ mole of helium gas is contained in a container at $S.T.P.$ The heat energy needed to double the pressure of the gas, keeping the volume constant (specific heat of the gas $ = 3\,J\,g{m^{ - 1}}\,{K^{ - 1}})$ is ...... $J$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of $2$ moles of helium gas (atomic mass $=4 \ amu$ ), and $1$ mole of argon gas (atomic mass $=40 \ amu$ ) is kept at $300 \ K$ in a container. The ratio of the rms speeds $\left(\frac{\left. v _{ mms } \text { (helium }\right)}{ v _{\text {rms }} \text { (argon) }}\right)$ is :View Solution
- 2At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 3View SolutionBoyle's law holds for an ideal gas during
- 4The temperature of a gas at pressure $P$ and volume $V$ is $27°C.$ Keeping its volume constant if its temperature is raised to $927°C,$ then its pressure will beView Solution
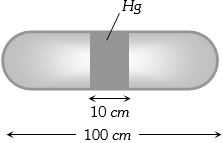
- 5A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 6If the ratio of vapour density for hydrogen and oxygen is $\frac{1}{{16}}$, then under constant pressure the ratio of their rms velocities will beView Solution
- 7The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
- 8In the two vessels of same volume, atomic hydrogen and helium at pressure $1\, atm$ and $2\, atm$ are filled. If temperature of both the samples is same, then average speed of hydrogen atoms $ < {C_H} > $ will be related to that of helium $ < {C_{He}} > $ asView Solution
- 9Two gases occupy two containers $A$ and $B$ the gas in $A$, of volume $0.10\,m ^3$, exerts a pressure of $1.40\,MPa$ and that in $B$ of volume $0.15 m ^3$ exerts a pressure $0.7\,MPa$. The two containers are united by a tube of negligible volume and the gases are allowed to intermingle. Then if the temperature remains constant, the final pressure in the container will be (in MPa)View Solution
- 10The equation for an ideal gas is $PV = RT,$ where $V$ represents the volume ofView Solution
