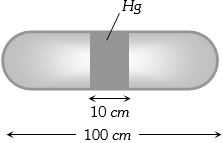
A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The number of air molecules per $\mathrm{cm}^3$ increased from $3 \times 10^{19}$ to $12 \times 10^{19}$. The ratio of collision frequency of air molecules before and after the increase in number respectively is $.........$View Solution
- 2In the relation $n = \frac{{PV}}{{RT}},n = $View Solution
- 3View SolutionInternal forces acting in a system of particle can change
- 4View SolutionOn absolute temperature, the kinetic energy of the molecules
- 5A container holds $10^{26} molecules/m^3$ each of mass $3 \times 10^{-27}\,\,kg$. Assume that $1/6$ of the molecules move with velocity $2000 \,\,m/s$ directly towards one wall of the container while the remaining $5/6$ of the molecules move either away from the wall or in perpendicular direction, and all collisions of the molecules with the wall are elasticView Solution
- 6The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$View Solution
- 7Consider the following statements for air molecules in an air tight container :View Solution
$(I)$ The average speed of molecules is larger than root mean square speed.
$(II)$ Mean free path of molecules is larger than the mean distance between molecules.
$(III)$ Mean free path of molecules increases with temperature.
$(IV)$ The rms speed of nitrogen is smaller than oxygen molecule.
Which of the above statements are correct?
- 8At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 9At a pressure of $24 \times {10^5}\,dyne/c{m^2}$, the volume of ${O_2}$ is $10\, litre$ and mass is $20\, gm.$ The $r.m.s.$ velocity will be....... $m/sec$View Solution
- 10If the degree of freedom of a gas are $f,$ then the ratio of two specific heats ${C_P}/{C_V}$ is given byView Solution
