Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 2A real gas within a closed chamber at $27^{\circ} \mathrm{C}$ undergoes the cyclic process as shown in figure. The gas obeys $P V^3=\mathrm{RT}$ equation for the path $A$ to $B$. The net work done in the complete cycle is (assuming $R=8 \mathrm{~J} / \mathrm{molK}$ ):View Solution
- 3View SolutionA given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an
- 4$Assertion :$ Reversible systems are difficult to find in real world.View Solution
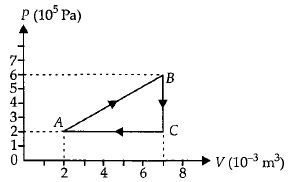
$Reason :$ Most processes are dissipative in nature. - 5A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 6View SolutionIn a cyclic process, the internal energy of the gas
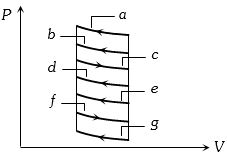
- 7The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
- 8$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
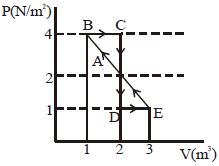
- 9One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
- 10View SolutionIn a cyclic process, work done by the system is
